The maximum flexibility and versatility for the best quality finished products
Long-lasting stability
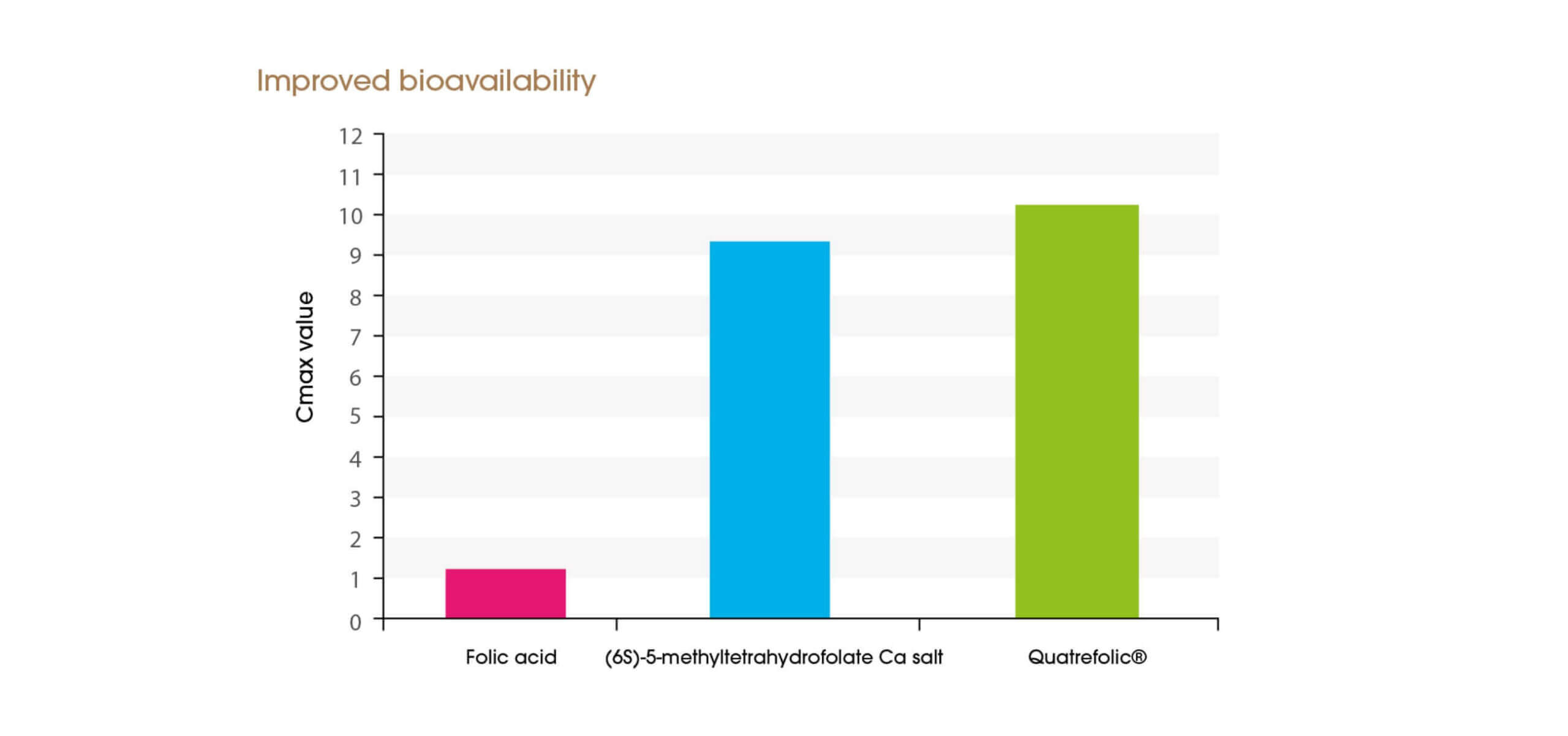
Quatrefolic® shows a long-lasting chemical stability at both room and refrigerated conditions, guaranteeing purity and ensuring easy handling and storage. Stability at room temperature might be a useful indicator to predict shelf life especially if there is potential for combining the Quatrefolic® with other components such as vitamins and minerals.
Stability studies of Quatrefolic® have been performed according to ICH (International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use) guidelines. Entering into its third decade of activity, the Global Quality Expert Working Group of ICH undertakes to rationalise and harmonise regulation in the world (the USA, Europe and Japan) to ensure that safe, effective, and high quality medicines are developed and registered in the most resource-efficient manner.
Gnosis by Lesaffre has deliberately chosen to carry out stability tests of Quatrefolic® according to the most strict guidelines, specifically required for pharmaceutical ingredients guaranteeing undependable and reliable criteria to claim the long-lasting chemical stability to its clients.